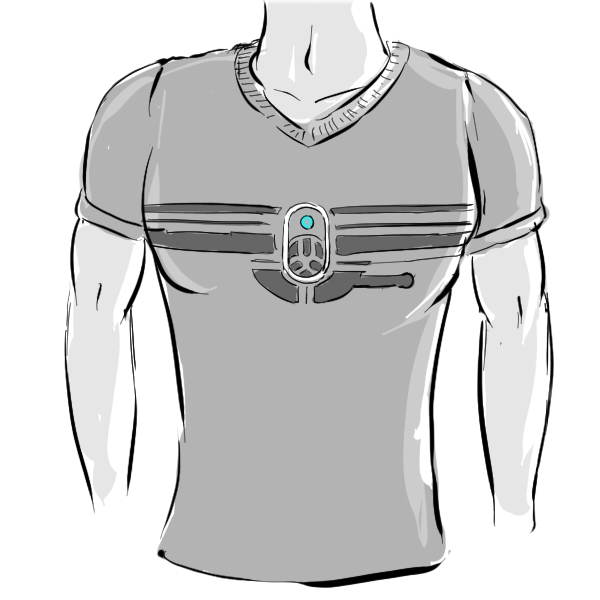
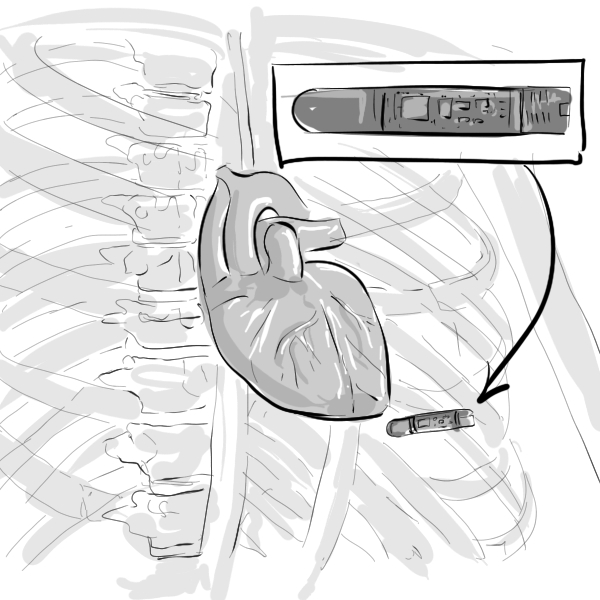
TODAY THERE IS AN EMPHASIS TO IMPROVE QUALITY OF LIFE (QOL) AMONG HF PATIENTS WHILE MANAGING SYMPTOMS, DEFINING THE CLINICAL AND ECONOMIC GOALS.
26 M
HF patients worldwide
6M in U.S.
$ 108 B
Economic burden
$32B in U.S.
74 %
HF patients with
comorbidities
PARTNERSHIPS
We partner with medical device manufacturers to incorporate our technology.
Our team has decades of experience with hardware design, signal processing, algorithm development, and regulatory approval of medical devices.
Audicor technology is designed and developed under ISO 13485 QMS controls.
TEAM

Peter Bauer
PRESIDENT

Alan Andresen
VP ENGINEERING

Tim Wheeler
CHIEF TECHNOLOGY OFFICER

Patti Arand
DIRECTOR RESEARCH AND ALGORITHMS

Gregg Harris
DIRECTOR QUALITY AND REGULATORY